The Crystal16 is a medium throughput platform with up to 16 samples running at one time, in 4 independent temperature profiles from -20 to 150 °C. Each reactor is equipped with real time non-invasive transmissivity analytics allowing for live information about your samples at 1 ml scale. This results in a plethora of applications in a wide range of industries, including oil and gas, pharmaceuticals and polymers.
In this article we will cover publications in the past year that utilized Crystal16.

Crystal16 for studying primary and secondary nucleation rates
Sefcik et al [1] were able to outline a workflow for rapid assessment of primary and secondary nucleation rates, as well as crystal growth rates. Initial solubility screening was conducted on glycine in deionized water, which allowed them to construct solubility and metastable zone width curves (MSZW). From these curves, they could determine supersaturation, as well as identify suitable regions to study primary and secondary nucleation. Then, measurements done using the Crystalline instrument allowed them to quantify nucleation and crystal growth rates in seeded and unseeded samples to quantitatively assess crystallization behavior. This revealed a clearly defined quantitative relationship between nucleation and growth kinetics determined at given crystallization conditions. These relationships are based on power law, dependent on supersaturation level that both growth and nucleation kinetics tend to follow.
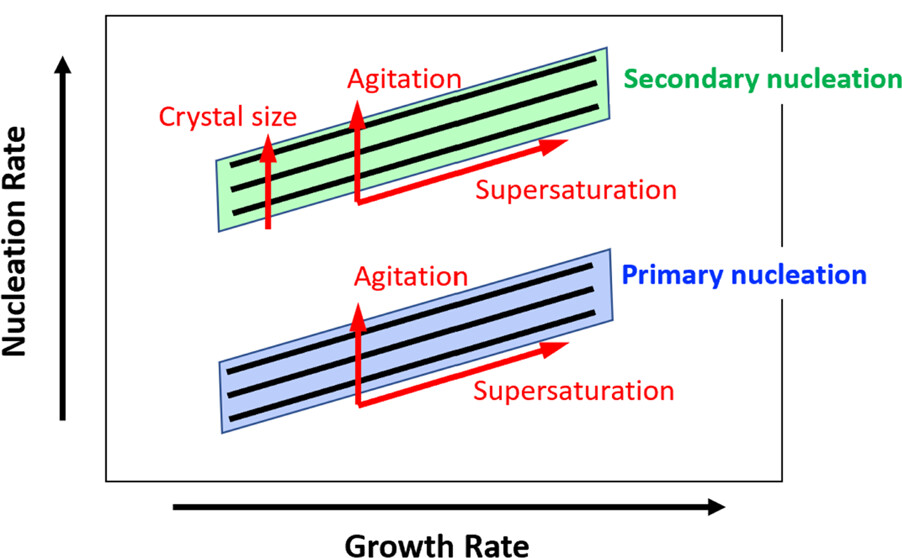
Figure 1: Schematic of crystallization behavior assessment showing expected dependencies of nucleation and growth rates on the supersaturation (Sefcik et al.)
Crystal16 for investigating the influence of solvent selection
Roberts et al [2] used the Crystal16 in their work investigating the influence of solution environment on the solution thermodynamics, crystallizability and nucleation rates of tolfenamic acid Form II. This was done by performing temperature cycling in the Crystal16 between -20 °C and 50 °C in 5 solvents (methanol, ethanol, isopropanol, toluene and acetonitrile) at 5 different heating and cooling rates measuring dissolution and nucleation points. Then, the solids were isolated and analysed by FT-IR to determine solid form. Next, modelling was done using the data and the results showed that the crystallizability was consistent with respect to the diffusion coefficient and mass transfer. Where IPA has a 10-times higher diffusion coefficient than ACN, this was reflected in their relative nucleation rates.
Crystal16 for studying ice nucleation
2023 saw interesting research into ice nucleation using the Crystal16. The process of ice nucleation is of interest for biopharmaceuticals, which commonly have limited stability in aqueous solution and require freezing in order to meet shelf-life requirements. However, as nucleation is a stochastic process, it presents a major challenge in freezing processes of biopharmaceuticals in vials. Deck et al [3] utilized the Crystal16 medium throughput design to generate ice nucleation times with temperature cycling samples with different ramp rates. Using optional thermocouples placed in each vial, nucleation was detected by the temperature change from the crystallization exotherm during cooling. Using the exotherm and nucleation rates, the researchers were able to validate experimental parameters and develop a model for the freezing process design. They followed this up later in the year using the same detection method in the Crystal16 to investigate the effects solution composition has on the nucleation kinetics of ice [4]. In these experiments, the researchers varied the sucrose composition. It was found that the stochastic ice nucleation kinetics are independent of the nature and concentration of solute. Additionally, the research team found that a single set of parameters can quantitatively describe the nucleation behavior of all solutions tested.
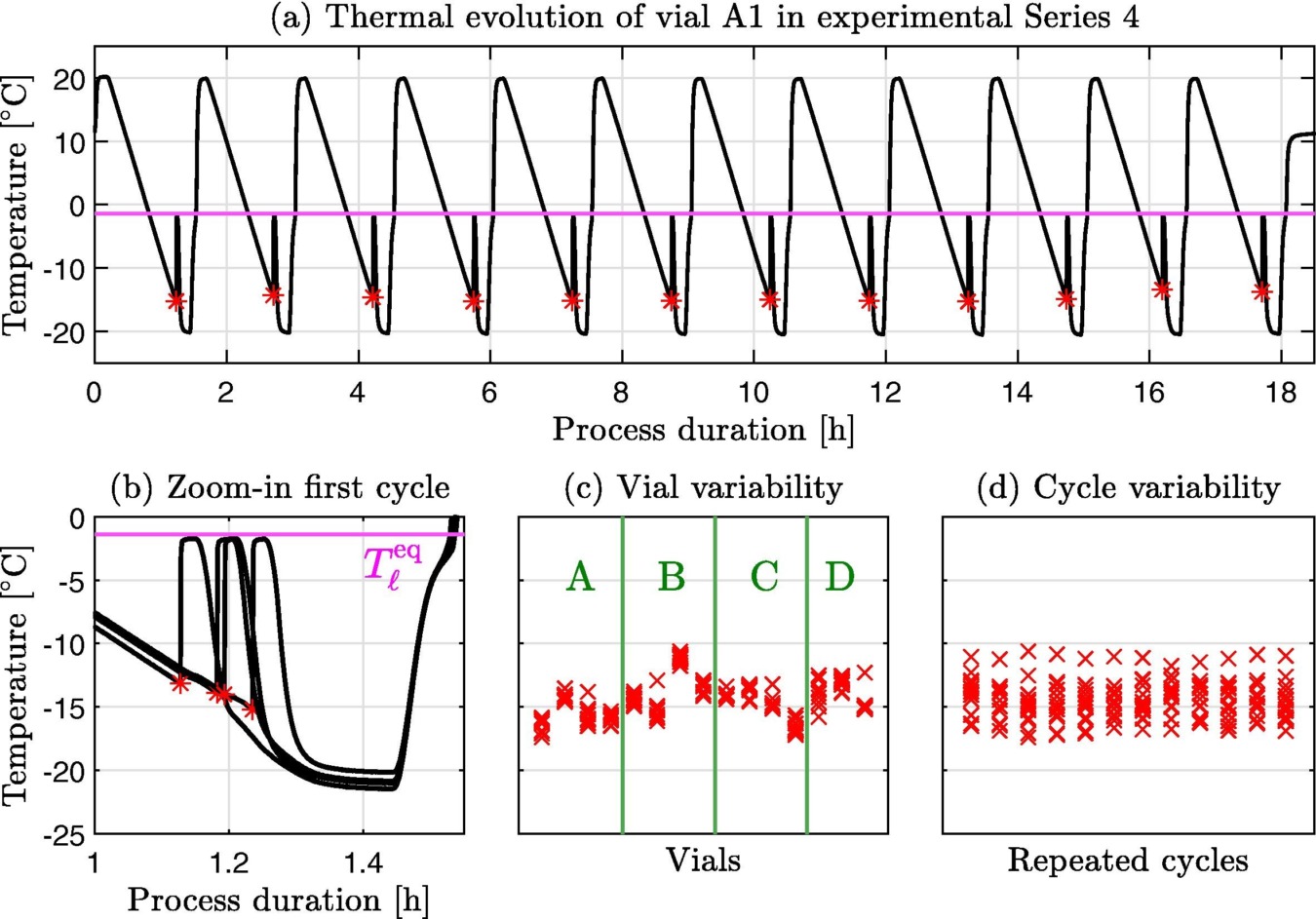
Figure 2: The thermal evolution of the four vials in reactor (Deck et al.)
Crystal16 for assessing solubility and the solid form landscape
Flavonoids are naturally occurring substances used in the nutraceutical and food industries. Similarly, to other small molecules, the solid forms can exhibit different physiochemical properties, thus impacting the compound’s performance. Klitou et al [5] investigated the solid form landscape of quercetin, which is an anti-inflammatory. Several solid forms were found: a methanol (QME) and ethanol (QE) solvate, a dihydrate (QH), and 2 anhydrous forms (QA and QA2). Solubility of the solid forms in IPA were assessed using the Crystal16 as part of characterization to understand the solid form landscape. From the information collected they were able to create a form diagram covering newly discovered forms as well as previously known forms.
Crystal16 for studying polymers
The Crystal16 has also been used in the study of polymers, such as in the work of Amriheasari et al [6]. The aim of their work was to develop a robust solubility methodology, with the goal of moving towards high throughput measurements. The researchers utilized a polythermal method between 10 °C and 60 °C with varying ramp rates, stir rates, as well as holding and cycling times, for 3 systems: polypropylene in toluene, polyethylene glycol in toluene and polycaprolactone in DMF. The results provided a strong foundation for designing dissolution measurements, thus improving the quality and collection speed of polymer solubility.
Crystal16 for conducting crystallization in crude oil research
Crude oil research has also been conducted on the Crystal16 by Ali et al [7]. Asphaltenes and waxes are two components of crude oil that cause flow issues due to their formation and crystallization. Therefore, it is of interest to investigate the effect their concentration has on the temperature this occurs at. The wax dissolution and crystallization were studied using a polythermal method of two different concentrations of asphaltenes with different ramp rates. The results were then interpreted with additional analysis by colloidal probe atomic force microscopy. The study highlighted the importance of asphaltene-asphaltene interactions modifying the bulk properties.
References
- Andrew Cashmore, Russell Miller, Hikaru Jolliffe, Cameron J. Brown, Mei Lee, Mark D. Haw, and Jan Sefcik. Crystal Growth & Design 2023 23 (7), 4779-4790, DOI: 10.1021/acs.cgd.2c01406
- Yu Liu, Cai Y. Ma, Junbo Gong, and Kevin J. Roberts Crystal Growth & Design 2023 23 (8), 5846-5859, DOI: 10.1021/acs.cgd.3c00450
- Leif-Thore Deck, Marco Mazzotti, Characterizing and measuring the ice nucleation kinetics of aqueous solutions in vials, Chemical Engineering Science, Volume 272, 2023, 118531, https://doi.org/10.1016/j.ces.2023.118531.
- Leif-Thore Deck, Lisanne Wittenberg, and Marco Mazzotti The Journal of Physical Chemistry Letters 2023 14 (26), 5993-6000, DOI: 10.1021/acs.jpclett.3c01371
- Panayiotis Klitou, Emmanuele Parisi, Simone Bordignon, Federica Bravetti, Ian Rosbottom, Marzia Dell’Aera, Corrado Cuocci, Michele R. Chierotti, Angela Altomare, and Elena Simone, Crystal Growth & Design 2023 23 (8), 6034-6045, DOI: 10.1021/acs.cgd.3c00584
- Mona Amrihesari, Amari Murry, Blair Brettmann, Towards standardized polymer solubility measurements using a parallel crystallizer, Polymer, Volume 278, 2023, 125983, https://doi.org/10.1016/j.polymer.2023.125983.
- Abdulraouf Ali, Ghinwa Yaghy, Laksha Parameswaran, Chris S. Hodges, Thibaut V.J. Charpentier, Simon D. Connell, Kevin J. Roberts, and David Harbottle, Energy & Fuels 2024 38 (1), 117-126 DOI: 10.1021/acs.energyfuels.3c02672
See how Crystal16 can help your research
Explore the instrument's multiple applications to enhance your research.
Book a demo with our experts!